In October 2021, the FDA formally proposed a rule to establish the new OTC Hearing Aids category. Finalized in August 2022, the final rule allows Hearing Aids within this category to be sold directly to consumers in stores or online without a medical exam or a fitting by an audiologist.
The published background of OTC hearing aid rule.
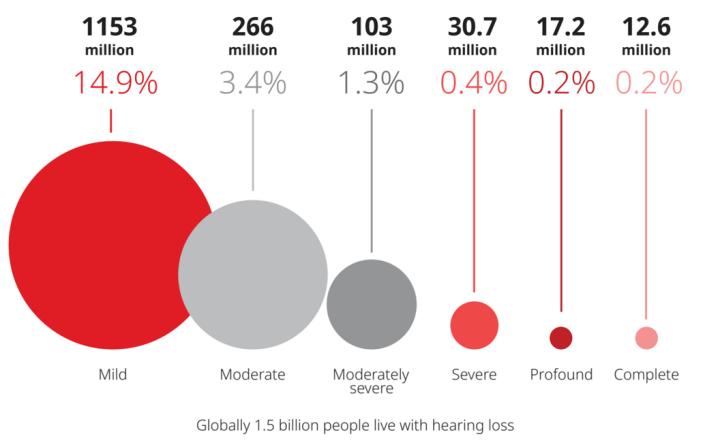
The world’s 1.5 billion people have hearing loss, and the wearing rate in China is less than 5%. According to US media sources, 37.5 million adults have hearing problems in the United States alone, and there are currently less than one-fifth of people who can use hearing aids. Looking at the global number is even more shocking. According to WHO (World Health Organization) statistics, 1.5 billion people around the world suffer from hearing loss(as following figure). The wear rate of hearing aid in China is far lower than the US level, even less than 5%.
The meaning the OTC hearing aid sells in our daily life.

In the United States, buying traditional hearing aids requires a doctor. This specification is to place the sales and use of hearing aid under relatively strict supervision to ensure the safety and effectiveness of users in the process of using hearing aids. But at the same time, it also brings the market closed, also the famous brand hearing aids are hard to afford for most people.
The OTC Act will allow it to be sold directly to mild to moderate listeners through the store or the Internet without the need for doctors or listeners, instead of doctors or listeners. prescription.
This move not only allows the hearing to be more convenient to buy hearing aid products but also introduces more manufacturers to increase market competition by reducing the threshold of sales channels.